Florida grand jury investigating COVID-19 vaccines finds no evidence of criminal activity
A statewide grand jury convened at the request of Florida Gov. Ron DeSantis to investigate “any and all wrongdoing" concerning COVID-19 vaccines did not find any evidence of criminal activity, according to a report unsealed on Tuesday.
A statewide grand jury convened at the request of Florida Gov. Ron DeSantis to investigate “any and all wrongdoing" concerning COVID-19 vaccines did not find any evidence of criminal activity, according to a report unsealed on Tuesday.
US health officials propose once-a-year COVID-19 vaccines for most Americans
The proposal comes as boosters have become a hard sell.
The proposal comes as boosters have become a hard sell.
Florida Supreme Court OKs DeSantis' request for grand jury investigation into COVID-19 vaccines
The Florida Supreme Court has approved Gov. Ron DeSantis' request for a grand jury to investigate any alleged wrongdoing related to COVID-19 vaccines. The statewide grand jury will be empaneled in Hillsborough County, and will meet for one year.
The Florida Supreme Court has approved Gov. Ron DeSantis' request for a grand jury to investigate any alleged wrongdoing related to COVID-19 vaccines. The statewide grand jury will be empaneled in Hillsborough County, and will meet for one year.
Fauci urges Americans — one last time — to get the updated COVID-19 booster
Dr. Fauci, who is just weeks away from retirement, had a consistent, familiar message for Americans during his last time at the podium — get your COVID shot.
Dr. Fauci, who is just weeks away from retirement, had a consistent, familiar message for Americans during his last time at the podium — get your COVID shot.
Florida Gov. Ron DeSantis says there will be no children's COVID vaccine mandate
Florida Surgeon General Joseph Ladapo has come out against routine COVID-19 vaccines for children.
Florida Surgeon General Joseph Ladapo has come out against routine COVID-19 vaccines for children.
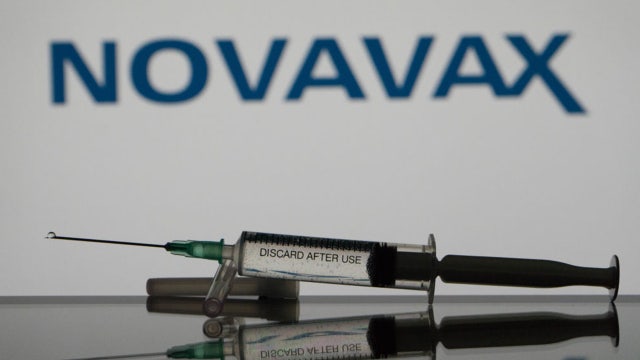
FDA OKs Novavax COVID-19 booster dose
The Food and Drug Administration has authorized a booster dose of the COVID-19 vaccine made by Novavax.
The Food and Drug Administration has authorized a booster dose of the COVID-19 vaccine made by Novavax.
White House: Get new COVID booster by Halloween
The White House is warning of a "challenging" virus season ahead and urging eligible Americans to get the updated COVID-19 boosters by Halloween to have maximum protection against the coronavirus by Thanksgiving and the holidays
The White House is warning of a "challenging" virus season ahead and urging eligible Americans to get the updated COVID-19 boosters by Halloween to have maximum protection against the coronavirus by Thanksgiving and the holidays
Should I get the new COVID booster and flu shot at the same time? Your questions, answered
With flu season approaching in the U.S., as well as the availability of the new omicron-designed COVID-19 booster for everyone 12 and up, here are some answers to common questions about the shots.
With flu season approaching in the U.S., as well as the availability of the new omicron-designed COVID-19 booster for everyone 12 and up, here are some answers to common questions about the shots.
Canada likely to drop vaccine requirement to enter, official says
Canada will likely drop the COVID-19 vaccine requirement for people who enter Canada by the end of September, an official familiar with the matter told The Associated Press.
Canada will likely drop the COVID-19 vaccine requirement for people who enter Canada by the end of September, an official familiar with the matter told The Associated Press.
Moderna sues Pfizer, BioNTech claiming companies violated patents with COVID vaccine
Moderna said in its complaint that Pfizer and BioNTech copied some “critical features” of its technology, including making the same "chemical modification” to their mRNA, which is used for protein production.
Moderna said in its complaint that Pfizer and BioNTech copied some “critical features” of its technology, including making the same "chemical modification” to their mRNA, which is used for protein production.
Osceola County Schools: Back-to-School free immunization events being hosted for students
Local schools across Osceola County are hosting free immunization events for students, including COVID-19 immunizations - Here’s everything you need to know.
Local schools across Osceola County are hosting free immunization events for students, including COVID-19 immunizations - Here’s everything you need to know.
NIH: COVID-19 booster-induced antibody response to omicron significantly decreases over time
Researchers found that while COVID-19 booster vaccinations create a strong antibody response against omicron initially, within three months post-boost, those levels decrease.
Researchers found that while COVID-19 booster vaccinations create a strong antibody response against omicron initially, within three months post-boost, those levels decrease.
CDC endorses more traditional Novavax COVID-19 vaccine for adults
Health officials say U.S. adults who haven’t gotten any COVID-19 shots yet should consider a new option from Novavax.
Health officials say U.S. adults who haven’t gotten any COVID-19 shots yet should consider a new option from Novavax.
US health officials grapple with whether to offer new COVID-19 booster shots this fall
"This is science at its toughest," FDA vaccine chief Dr. Peter Mark said of whether to offer modified COVID-19 booster shots this fall due to the changing coronavirus.
"This is science at its toughest," FDA vaccine chief Dr. Peter Mark said of whether to offer modified COVID-19 booster shots this fall due to the changing coronavirus.
V is for vaccine: Sesame Street's Elmo gets COVID-19 vaccine
Sesame Workshop, the nonprofit educational organization behind Sesame Street, announced that Elmo had gotten vaccinated in public service announcement posted to YouTube.
Sesame Workshop, the nonprofit educational organization behind Sesame Street, announced that Elmo had gotten vaccinated in public service announcement posted to YouTube.
FDA advisers recommend updating COVID booster shots for fall
Some U.S. adults are a step closer to getting updated COVID-19 boosters this fall, as government advisers voted Tuesday that it’s time to tweak shots to better match the most recent virus variants.
Some U.S. adults are a step closer to getting updated COVID-19 boosters this fall, as government advisers voted Tuesday that it’s time to tweak shots to better match the most recent virus variants.
COVID-19 vaccine: CDC approves 1st shots for US kids under 5
With the CDC's final sign-off, roughly 18 million children under 5 will be eligible for COVID-19 shots beginning next week.
With the CDC's final sign-off, roughly 18 million children under 5 will be eligible for COVID-19 shots beginning next week.
COVID-19 vaccine: FDA authorizes 1st shots for kids under 5; CDC review next
The CDC recommends how to use vaccines and its vaccine advisers are set to discuss the shots for the youngest kids Friday and vote on Saturday.
The CDC recommends how to use vaccines and its vaccine advisers are set to discuss the shots for the youngest kids Friday and vote on Saturday.
FDA panel endorses Moderna's COVID vaccine for teens, school-age kids
An FDA advisory panel endorsed on Tuesday Moderna's COVID-19 vaccine for teenagers and school-aged children.
An FDA advisory panel endorsed on Tuesday Moderna's COVID-19 vaccine for teenagers and school-aged children.
US: Pfizer COVID-19 shot appears effective for kids under 5
Federal health officials said Sunday that kid-sized doses of Pfizer’s COVID-19 vaccines appear to be safe and effective for kids under 5, a key step toward a long-awaited decision to begin vaccinating the youngest American children.
Federal health officials said Sunday that kid-sized doses of Pfizer’s COVID-19 vaccines appear to be safe and effective for kids under 5, a key step toward a long-awaited decision to begin vaccinating the youngest American children.